 The rate law for a chemical reaction describes the relationship between the rate of the reaction and the concentrations of the reactants. The reaction is called the 'iodine clock' because the appearance of the blue-black color of iodine indicates the completion of the reaction. 
The reaction produces iodine (I2) and water (H2O) as products. The iodine clock reaction is a chemical reaction that involves the oxidation of iodide ions (I-) by a strong oxidizing agent, such as hydrogen peroxide (H2O2), in the presence of an acid. 
In this blog post, we will delve into the rate law for iodine clock reaction, a classic example used to study chemical kinetics. From the rusting of iron to the combustion of fuels, understanding the factors that influence reaction rates is crucial in various fields of science and industry. 2002, 79, p 41.Understanding the Rate Law for Iodine Clock ReactionĬhemical reactions are fundamental processes that occur all around us. Additionally, it helps students understand the importance of green chemistry at an early stage in their learning career. Putting green chemistry principles into practice by eliminating the use of toxic chemicals and hazardous waste is an excellent way to help mitigate these challenges. The transportation of reagents and the safe disposal of chemicals are often two of the biggest challenges facing chemistry demonstrators. The difference now is that I could comfortably carry all the required chemicals on public transit and could rinse all of the waste from this demonstration safely down the sink. The same results are achieved – a dramatic colour change that gets students excited about chemistry. The first solution in the new procedure is made up of vitamin C powder dissolved in water and a few drops of tincture of iodine, while the second contains 3% hydrogen peroxide and laundry starch. I was able to purchase all the required reagents at Shoppers Drug Mart and the demonstration can be safely repeated by students at home. With the authors’ modifications, no concentrated acid or peroxide is needed. Luckily, I stumbled upon a very well written paper published in the Journal of Chemical Education where the authors managed to achieve the same demonstration using only consumer products. I began doing chemistry demonstrations at local high schools and quickly realized that this experiment would be challenging as it is not feasible for me to use some of these reagents both 20% hydrogen peroxide and concentrated sulfuric acid are corrosive and must be treated before disposal.Īll the reagents needed for this modified procedure of the iodine clock demonstration. Typically, one solution containing sodium bisulfite, potassium iodide, and soluble starch and a second solution containing 20% hydrogen peroxide and concentrated sulfuric acid are employed in the demonstration. While the reagents required for the iodine clock reaction can be easily found in a chemistry research lab, they are not necessarily the most user-friendly when it comes to doing this demonstration in a public setting. 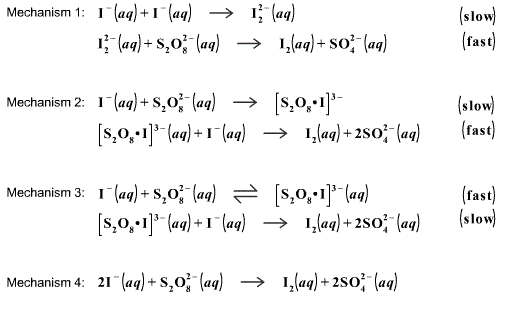
The first step is faster than the second step, thus no change is observed until all the reducing agents in the solution are consumed and step 1 cannot occur anymore, allowing step 2 to proceed to step 3. A number of similar procedures can be found online, yet the overall reaction always consists of the same three steps: 1) In the first step, iodine is reduced to iodide, 2) iodide then gets oxidized back to iodine, and finally 3) the iodine reacts with the starch contained in the mixture to give the deep blue colour. The time it takes for the colour change to take place can be altered by varying the concentration and temperature of the solutions. In addition to the dramatic colour change, this activity also serves to illustrate important concepts in reaction kinetics. This variation of the iodine clock reaction has the same dramatic colour change as the traditional demo! 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
